When I’m out in the ocean, I’m obsessed by everything I see. I’m the woman in the boat who’s got her fish guide out to look up the names of any unknown fish while throwing out fun facts about the creatures I’ve already identified. Yet so much ocean life is so small, I might miss them entirely. I’m talking about plankton—the tiny plants and animals that make up 90% of the mass of all marine life in the ocean and play an extremely important role in the health of our planet.
Love ocean content?
Enter your email and never miss an update
Plankton are the start of the ocean’s food chain. Phytoplankton (a.k.a. plant-type plankton) turn sunlight into energy via photosynthesis and are eaten by zooplankton (a.k.a. animal-type plankton), small fish and invertebrates. Zooplankton, in turn, become food for larger species like fish, whales or crustaceans. Phytoplankton also pump out oxygen and sequester carbon. Five hundred million years ago, a bloom of plankton created the breathable oxygen-rich atmosphere humans depend on today.
Despite being so crucial to life on this planet, plankton are best defined by their go-with-the-flow nature. An organism is considered plankton if it can’t swim or move against the forces of the ocean like currents and tides. That’s why the name comes from a Greek word meaning “drifter.” A vast diversity of life falls under the heading of plankton from krill to single-celled algae to the offspring of crabs to jellyfish. Each plankter (that’s actually the word for individual plankton) is unique, and today I’m going to introduce you to a couple of the wandering creatures.
Dinoflagellates
Dinoflagellates are among the most common type of plankton with more than 1,500 species living in our ocean. They are single-celled phytoplankton with flagella, thread like appendages that allow these microscopic organisms to move. These tiny organisms can be responsible for wonder and destruction in the ocean. If you’ve ever swum in a bioluminescent bay, the light you see is caused by dinoflagellates. The light may help distract or startle would-be predators. In addition, some species have symbiotic relationships, helping corals, jellyfish and other animals thrive.
On the flip side, dinoflagellates are largely responsible for deadly red tides. The term “red tide” comes from the pigment found in these tiny creatures and the color of the water when these blooms happen. When dinoflagellates grow uncontrollably, they can produce toxins and reduce the oxygen in the water leading to die-offs in the ocean. Red tides can cause health hazards for people on land and lead to the deaths of manatees, fish and other creatures in the ocean.
Baby Sea Creatures

My favorite activity growing up was tide pooling, but I never really thought about how many of the creatures I encountered ended up there in the first place. That’s because the offspring star fish, sea urchins, sea anemones, barnacles, crabs, even octopuses start their lives as plankton that look almost nothing like the creatures they will one day become. If you want to play the wildest game of “Where are they now?” try to match ocean creatures with their baby forms in this quiz from The American Museum of Natural History.
Siphonophores

There are about 175 different species of siphonophores, the most famous being the Portuguese Man-O’-War. Another notable siphonophore is the Apolemia, which may be the longest creature on Earth at 150 feet long. That’s about the length of 1.5 blue whales! Proof that not all plankton are the tiny creatures we commonly think of them to be.
While they may appear to be a single organism, they are actually a colony of individuals called zooids. Each zooid has a unique function in the colony. For instance, there are zooids that are responsible for swimming and moving around, while others specialize in digestion and providing food for the whole colony. There are even battle zooids that are always on the ready to inject toxins into prey.
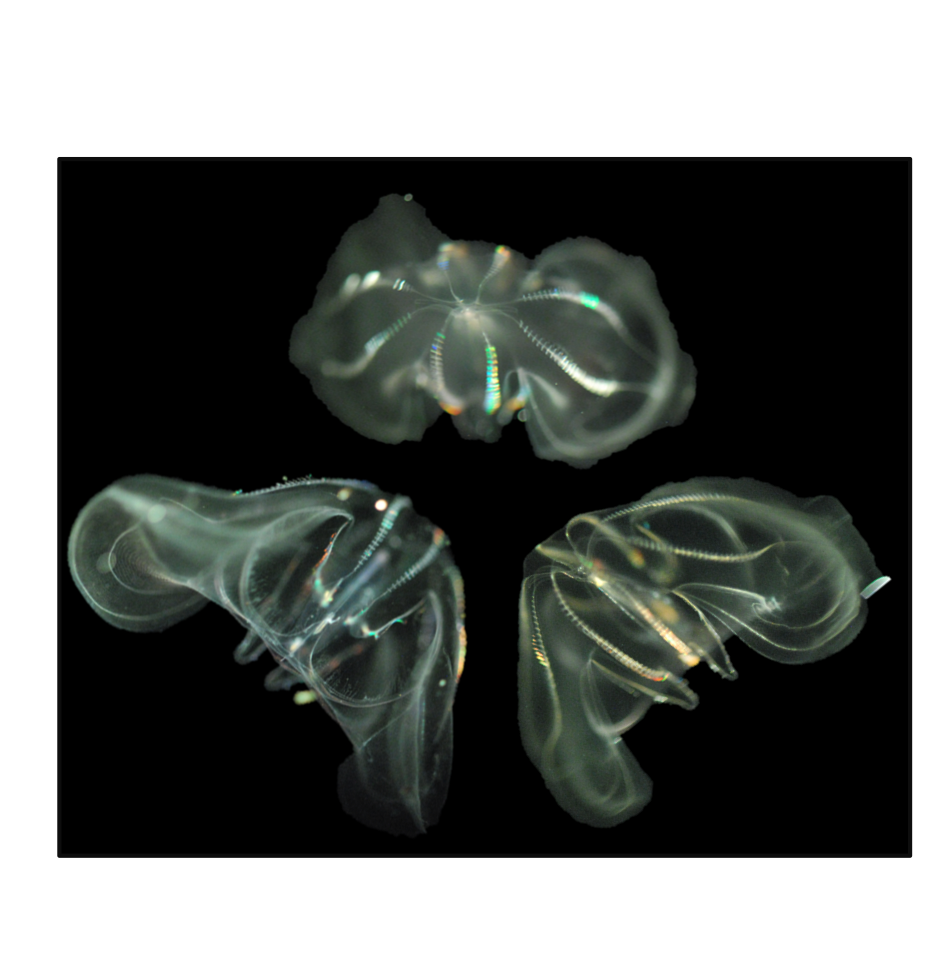
Comb Jellies

When you think of plankton, you often think of tiny animals, not beautiful otherworldly creatures like comb jellies. They are mesmerizing to watch because they produce a dazzling array of colors. This effect is caused by their cilia, which are like little hairs and are what give the comb jellies their name. These combs diffract light, breaking it up like a rain droplet or prism, to create rainbows.
Despite the similar name, comb jellies are not types of jellyfish. In fact, they could be descended from the Earth’s oldest animals. Recent research suggests that they could be the closest relatives of the first group to split off from our common ancestor around 500 million years ago. Comb jellies are pretty simple creatures without intestines, lungs or stomachs but they do have a nervous system that has puzzled scientists. Comb jellies have a nerve net that doesn’t have the usual synapses that humans and most other animals need for their neurons to communicate. Comb jellies could hold the answers to so many questions we have about how life evolved on Earth.
Diatoms

Diatoms are single-celled algae and among the largest groups of life on Earth. You can find diatoms anywhere it’s wet: freshwater, the ocean, even in moist soil. There are more than 100,000 species of diatoms and counting, as scientists find new types every year. What each diatom has in common is a cell wall made of silica, the same component we use to make glass, meaning they literally live in glass houses. Silica is also the main component of opals, and diatoms share some of that beauty, earning the name “jewels of the sea.” Diatoms are not only beautiful but are also powerful. They produce a significant amount of the air we breathe and are critical to marine ecosystems all over the world.
Some of the most amazing creatures on Earth are the ones we can’t easily see on the surface. Plankton play a critical role in sustaining life on this planet. Climate change is causing disruptions that we can see like intensifying storms, but it is also causing issues that we can’t see. That’s why it is so important that we all do what we can now to reduce polluting carbon emissions that are changing the ocean’s temperature, chemistry and function. Ocean Conservancy views climate change as the greatest challenge facing our planet today and is committed to finding ocean-based solutions to address it. Join us in taking action to combat climate change now.
The post Five Types of Plankton appeared first on Ocean Conservancy.
Ocean Acidification
Chemie: macht das Unsichtbare sichtbar
English version below
Wenn man an Chemie denkt, denkt man wahrscheinlich schnell an explodierende Gläser, ätzende Säuren und verrückte Professoren, aber nicht an den Ozean. Hier an Bord wird unsere Wissenschaftsteam auch von zwei chemischen Ozeanographen begleitet, Tobias Steinhoff und Kristin Kampen.
Den beiden habe ich die Frage gestellt, „Was findet ihr an der chemischen Ozeanografie spannend?“: Es ist unglaublich interessant, was es alles an unsichtbaren Prozessen im Meer gibt, die unser aller Leben beeinflussen: In der chemischen Ozeanographie untersuchen wir, wie sich chemische Bestandteile im Meer verhalten, z.B. wie sich gelöste Gase (wie CO₂ und Sauerstoff), Nährsalze (wie Nitrat und Phosphat), Spurenmetalle und organische Verbindungen im Meerwasser verhalten und verteilen. Der Ozean nimmt CO₂ auf, produziert Sauerstoff und transportiert Nährstoffe durch den Ozean und überall wirken chemische Prozesse mit. Diese Zusammenhänge zu verstehen ist Grundlage unserer Arbeit.
Auf unserer Ausfahrt in der Labradorsee nehmen sie Seewasserproben und extrahieren gelöstes organisches Material (DOM). Dies umfasst alle organischen Verbindungen, die im Meerwasser gelöst sind, also nicht als Partikel vorliegen. Das sind zum Beispiel Zucker, Aminosäuren, Fette und komplexere Moleküle, die aus abgestorbenen Organismen, Ausscheidungen von Meereslebewesen oder dem Abbau von Algen stammen. Als einer der größten Kohlenstoffspeicher spielt DOM eine zentrale Rolle im marinen Kohlenstoffkreislauf. Die Labradorsee ist eine der wichtigsten Regionen für die Bildung des North Atlantic Deep Water (NADW). Oberflächenwasser sinkt in die Tiefe und nimmt dabei DOM mit. Das NADW verteilt dieses Material dann über Jahrhunderte durch die Weltmeere und entzieht so der Atmosphäre langfristig Kohlenstoff. Zusätzlich werden kontinuierliche Messungen von pCO₂/O₂ im Oberflächenwasser während der Fahrt durchgeführt, um sich den Austausch von CO₂ zwischen Ozean und Atmosphäre anzuschauen. Viele Prozesse sind hierbei immer noch nicht vollständig verstanden, wie z.B. der Gasaustausch bei hohen Windgeschwindigkeiten.


Da es hier auf See, besonders in dieser Region, oft sehr stürmisch zugeht, ist kein Geheimnis und es geht natürlich besonders in einem Chemie Labor dann doch mal etwas zu Bruch. Wie läuft diese Arbeit also bei 11bft und 6 Meter Wellen ab. Wasserproben müssen meist innerhalb von 24 Stunden verarbeitet werden. Da kann man nicht immer Rücksicht auf die Wetterbedingungen nehmen. Einige Arbeiten werden immer noch nasschemisch gemacht und unter Einsatz von Glasmaterial. Sowohl das genaue Abmessen von Reagenzien als auch das Zusammenhalten der Glasware ist nicht immer einfach bei einem rollenden Schiff (und auch nicht immer erfolgreich). Man versucht zwar den doch dann plötzlichen Bewegungen des Schiffes entgegenzuwirken und alle Proben Behälter, Kisten und Flaschen zu sichern. Man wird aber dann doch mal von einem umkippenden Mülleimer überrascht und die noch neu verpackten Plastikröhrchen oder andere Fliegengewichte im Regal finden bei der einen oder anderen Welle ihren Weg auf die gegenüberliegende Seite im Labor. Dazu kommt, dass beim Arbeiten mit chemischen Stoffen und Proben doch des Öfteren beide Hände für die Arbeit gebraucht werden. Wird man dann allerdings von einer Welle überrascht, erfordert das Festhalten mit der dritten Hand (Fuß falls man schnell genug ist), einiges an Bauchmuskeln.

Foto: Julia Pelle
Das Besondere an der Arbeit auf See ist, dass man neben der alltäglichen Schreibtischarbeit auch praktisch arbeiten kann. Dabei ist man auf die enge Zusammenarbeit mit seinen Kollegen angewiesen und lernt sie dabei viel besser kennen. Zusätzlich sind auch viele andere Forschungsbereiche mit an Bord, wodurch es einen spannenden Austausch zwischen den einzelnen Gruppen gibt.
Zum Schluss hier noch ein kleiner Tipp am Rande von unseren Chemikern und für deine erste Forschungsseereise: Laschen, laschen, laschen und immer ein Ohr am Bordfunk: Der Arbeitsplan ist bei den Wetterbedingungen eher ein Vorschlag und kann sich stündlich ändern (die nächste CTD Station ist immer um die Ecke).
Chemistry: Making the Invisible Visible
When you think of chemistry, you probably quickly imagine exploding glassware, corrosive acids, and crazy professors, but not the ocean. Here on board, our scientific team is also accompanied by two chemical oceanographers, Tobias Steinhoff und Kristin Kampen.
I asked them the question: “What do you find exciting about chemical oceanography?”
“It is incredibly fascinating how many invisible processes exist in the ocean that influence all of our lives. In chemical oceanography, we study the fate of various chemical components in the ocean: for example, how dissolved gases (such as CO₂ and oxygen), nutrients (such as nitrate and phosphate), trace metals, and organic compounds behave and are distributed in seawater. The ocean absorbs CO₂, produces oxygen, and transports nutrients through complex cycles, including chemical processes. Understanding these relationships forms the basis of our work.”
During our expedition in the Labrador Sea, they collect seawater samples and extract dissolved organic material (DOM). This includes all compounds dissolved in seawater, meaning they are not present as particles. Examples include sugars, amino acids, fats, and more complex molecules that originate from dead organisms, excretions from marine life, or the breakdown of algae. As one of the largest carbon reservoirs, DOM plays a central role in the marine carbon cycle.


The Labrador Sea is one of the most important regions for the formation of North Atlantic Deep Water (NADW). Surface water sinks into the depths, carrying DOM with it. NADW then distributes this material throughout the world’s oceans over centuries, thereby removing carbon from the atmosphere over the long term. In addition, continuous measurements of pCO₂ and O₂ in surface water are taken during the voyage to study the exchange of CO₂ between the ocean and the atmosphere. Many processes involved are still not fully understood, such as gas exchange under high wind speeds.
It is no secret that conditions at sea especially in this region are often very stormy, and in a chemistry lab, things can occasionally break. So how does this work at 11 Beaufort and 6-meter waves? Water samples usually need to be processed within 24 hours, so you cannot always take weather conditions into account. Some work is still done using wet chemistry and glass equipment. Accurately measuring reagents and holding glassware steady is not always easy on a rolling ship (and not always successful). Although efforts are made to counteract sudden ship movements and to secure all sample containers, boxes, and bottles, you may still be caught off guard by a tipping trash bin, and newly packaged plastic tubes or other lightweight items can suddenly fly across the lab with the next wave.
On top of that, when working with chemicals and samples, both hands are often needed. If a wave hits unexpectedly, holding on with a “third hand” (your foot, if you are quick enough) requires quite a bit of core strength. What makes working at sea special is that, alongside everyday desk work, you can also do hands-on work. This requires close cooperation with colleagues, allowing you to get to know them much better. In addition, many other research disciplines are on board, which creates exciting exchanges between different groups.
Finally, here is a small tip from our chemists for your first research expedition: strap everything down, strap everything down, strap everything down and always keep one ear on the ship’s radio. The work schedule is more of a suggestion under these weather conditions and can change hourly (the next CTD station is always just around the corner).
Ocean Acidification
Between Storms and Science: Easter in the Labrador Sea (04.04.26–13.04.26)
Between all the scientific work, we celebrated Easter on board, although the weather had other plans for us. Due to rough conditions, we weren’t able to carry out any CTD casts.

Easter itself was spent in a mix of rest and small celebrations. Some of us enjoyed a long Easter breakfast with traditional Easter bread, while others took the opportunity to sleep in. In the evening, we gathered with both crew and scientists for a small celebration. The ship’s cook even organized a quiz, and those who answered correctly were rewarded with Easter chocolate.
The next day, the weather improved, and we began early with the recovery of K1, a 3,495-meter-long mooring in the middle of the Labrador Sea.
We joined the nautical officers on the bridge before sunrise to search for it. Fortunately, K1 has a floating buoy with a light, so we were able to spot it even in the dark. The actual recovery started at first light, and it began to snow while we were working.


Amid all the CTDs and mooring operations, there was also a personal highlight: my (Sarah’s) birthday. Although I’ve spent birthdays away from home before, this one felt especially unique, being so far out at sea, with only limited internet contact.
Normally, I work the 4-8 shift, but my incredibly kind shift team gave me the morning off. That meant I could sleep in and even find time to call family and friends back home. In the afternoon, I was surprised with my favourite cake, baked by Julia.
Our work continued with the mooring array at 53°N, which consists of seven moorings. So far, we have recovered five (K7, K8, K9, DSOW1 and DSOW2), and three of them have already been redeployed (K7, K8 and DSOW1,).
Deploying K7 turned out to be particularly tricky. On our first attempt, sea ice drifted toward us faster than expected, forcing us to recover nearly half of the mooring again. While the ship itself can handle drifting ice, deploying a mooring is much more delicate: a long cable with instruments and floats is released behind the ship before the anchor is dropped, allowing the system to sink into place.
Two days later, we tried again and this time, the deployment was successful.

Afterwards, we moved closer to the sea ice, which was a highlight for many of us. Seeing the ice up close and even spotting a seal swimming nearby, made the experience unforgettable.


Due to the continuing harsh weather, the decision was made to return to K1 and make use of an upcoming weather window for deployment the following day.
German:
Zwischen Stürmen und Wissenschaft: Ostern in der Labradorsee (04.04.26 – 13.04.26)
Zwischen all der wissenschaftlichen Arbeit haben wir Ostern an Bord gefeiert, auch wenn das Wetter andere Pläne für uns hatte. Aufgrund der rauen Bedingungen konnten wir keine CTD-Messungen durchführen (Messungen von Leitfähigkeit, Temperatur und Tiefe im Ozean).

Ostern selbst war eine Mischung aus Erholung und kleinen Feierlichkeiten. Einige von uns genossen ein ausgedehntes Osterfrühstück mit traditionellem Osterbrot, während andere die Gelegenheit nutzten, etwas länger zu schlafen. Am Abend kamen Crew und Wissenschaftler*innen zu einer kleinen Feier zusammen. Der Koch organisierte sogar ein Quiz, und wer die Fragen richtig beantwortete, wurde mit Oster-Schokolade belohnt.
Am nächsten Tag besserte sich das Wetter, und wir begannen früh mit der Bergung von K1, einer 3.495 Meter langen Verankerung mitten in der Labradorsee. (Eine Verankerung ist eine lange, am Meeresboden befestigter Draht, der mit Instrumenten ausgestattet ist, um über längere Zeit Ozeandaten zu messen.)
Noch vor Sonnenaufgang gingen wir mit den nautischen Offizieren auf die Brücke, um nach ihr Ausschau zu halten. Glücklicherweise verfügt K1 über eine schwimmende Boje mit Licht, sodass wir sie bereits im Dunkeln entdecken konnten. Die eigentliche Bergung begann bei Tagesanbruch und es begann sogar zu schneien.


Zwischen all den CTD-Einsätzen und Verankerungsarbeiten gab es auch ein persönliches Highlight: meinen (Sarahs) Geburtstag. Obwohl ich schon öfter Geburtstage fernab von zu Hause verbracht habe, war dieser besonders, so weit draußen auf dem Meer und mit nur eingeschränktem Internetkontakt.
Normalerweise arbeite ich in der 4-8 Uhr Schicht, aber mein unglaublich nettes Schichtteam hat mir den Morgendienst freigegeben. So konnte ich etwas länger schlafen und hatte sogar Zeit, mit Familie und Freunden zu Hause zu telefonieren. Am Nachmittag wurde ich dann noch mit meinem Lieblingskuchen überrascht, den Julia für mich gebacken hat.
Unsere Arbeit ging weiter mit dem Verankerungs-Array bei 53°, das aus sieben Verankerungen besteht. Bisher haben wir fünf geborgen (DSOW1, DSOW2, K7, K8 und K9), von denen drei bereits wieder ausgebracht wurden (DSOW1, K7 und K8).
Das Ausbringen von K7 erwies sich als besonders schwierig. Beim ersten Versuch trieb das Meereis schneller auf uns zu als erwartet, sodass wir fast die Hälfte der Verankerung wieder einholen mussten. Obwohl das Schiff selbst gut durch treibendes Eis navigieren kann, ist das Ausbringen einer Verankerung deutlich anspruchsvoller: Dabei wird ein langer Draht mit Messinstrumenten und Auftriebskörpern hinter dem Schiff ausgesetzt, bevor am Ende der Anker gelöst wird und das gesamte System absinkt.
Zwei Tage später versuchten wir es erneut, diesmal mit Erfolg.

Anschließend fuhren wir näher an das Meereis heran, was für viele von uns ein besonderes Highlight war. Das Eis aus nächster Nähe zu sehen und sogar eine Robbe in der Nähe schwimmen zu beobachten, machte das Erlebnis unvergesslich.


Aufgrund der weiterhin rauen Wetterbedingungen wurde schließlich entschieden, zu K1 zurückzukehren, um ein bevorstehendes Wetterfenster für die Ausbringung am nächsten Tag zu nutzen.
Between Storms and Science: Easter in the Labrador Sea (04.04.26–13.04.26)
Ocean Acidification
Humans Just Flew Around the Moon This Week. But Would Babies Born There Ever Truly Feel Gravity? Ask Jellyfish Babies.
This week, NASA’s Artemis II crew made history by flying around the Moon and returning safely to Earth, the first human journey to the Moon’s vicinity in more than 50 years. It was a stunning reminder that humanity is no longer just dreaming about living beyond Earth. We are actively rehearsing for it.
And that leads to a much stranger, deeper question: even if one day we build skyscrapers on the Moon, raise families there, and turn space into a place to live, will babies born away from Earth develop a normal sense of gravity? Or will their bodies learn the universe differently?
To explore that question, NASA once turned to an unexpected stand-in for human babies: jellyfish babies. On the STS-40 mission, scientists sent thousands of tiny jellyfish polyps into space because jellyfish, like humans, rely on gravity-sensing structures to orient themselves. The experiment asked a simple but profound question: if a living body develops in microgravity, will it still know how to handle gravity later?
The answer was both fascinating and unsettling. The jellyfish developed in space in large numbers, but once back under Earth’s gravity, the ones that had developed in microgravity showed far more pulsing abnormalities than the Earth-grown controls. In other words, their bodies formed, but their sense of balance did not seem to work quite the same way.
That is why this old jellyfish experiment still matters today. Before we imagine lunar cities, schools, nurseries, and generations born off-world, we need to ask not only whether humans can survive in space, but whether developing there changes how the body understands something as basic as up, down, and movement. Jellyfish babies cannot tell us everything about human children, but they may have given us one of the first clues that life born beyond Earth might not come home unchanged.
Reference: https://nlsp.nasa.gov/view/lsdapub/lsda_experiment/0c10d660-6b12-573d-8c3b-e20e071aed3b
Image: GEOMAR, Sarah Uphoff
-
Greenhouse Gases9 months ago
Guest post: Why China is still building new coal – and when it might stop
-
Climate Change9 months ago
Guest post: Why China is still building new coal – and when it might stop
-

 Greenhouse Gases2 years ago
Greenhouse Gases2 years ago嘉宾来稿:满足中国增长的用电需求 光伏加储能“比新建煤电更实惠”
-
Climate Change2 years ago
Bill Discounting Climate Change in Florida’s Energy Policy Awaits DeSantis’ Approval
-

 Climate Change2 years ago
Climate Change2 years ago嘉宾来稿:满足中国增长的用电需求 光伏加储能“比新建煤电更实惠”
-

 Renewable Energy6 months ago
Renewable Energy6 months agoSending Progressive Philanthropist George Soros to Prison?
-

 Carbon Footprint2 years ago
Carbon Footprint2 years agoUS SEC’s Climate Disclosure Rules Spur Renewed Interest in Carbon Credits
-
Greenhouse Gases10 months ago
嘉宾来稿:探究火山喷发如何影响气候预测




